
THE SKIN AND MIND CONNECTION
Content for this white paper was derived from an advisory board meeting of experts in the mind-skin connection as well as from the literature. This white paper was supported by Erno Laszlo, Inc.
Advisory Board Participants and Affiliations
Cherie Ditre MD (University of Pennsylvania), Richard Fried MD, PhD (Private Practice), Marsha Gordon MD (Mount Sinai), John Koo, MD (University of California San Francisco), Ladan Mostaghini MD (University of Wisconsin), Even Rieder MD (NYU Grossman School of Medicine), Mary L. Stevenson (NYU Grossman School of Medicine), Richard Wortzel (University of Pennsylvania)
Introduction
The skin is an architectural marvel, designed by evolution to be both protective and resilient. Pliable yet tough, the skin combats environmental insults through a program of continuous self-renewal that maintains its function. However, like all other organs of the human body, the skin is susceptible to both intrinsic and extrinsic factors that, over time, cause not only skin conditions, but also the visible and invisible signs of aging. The steady deterioration of the dermal and epidermal layers of the skin with age is caused by a combination of factors including reduced cellular proliferation and collagen synthesis, extracellular matrix remodeling, and changes in epidermal morphology (Crane 2015).
The skin protects the body’s contents through a series of chemical, mechanical, and biologic barriers. The chemical barriers of the skin include antimicrobial substances and melanin. The skin is host to abundant bacterial populations, the composition of which is modulated substances secreted by the skin. The low pH of the skin—referred to as the acid mantle—inhibits the proliferation of bacteria. At the same time, dermicidin and antibacterial substances in sebum kill bacteria directly, and skin cells also secrete natural antibiotics, the defensins, that perforate and kill bacteria. Wounded skin releases cathelicidins—a protective antibacterial peptide that helps prevent infection by group A streptococcus bacteria. The physical barrier of the skin largely arises from the structure of the stratum corneum, which consists of multiple layers of dead, flattened cells and glycolipids that together defend against mechanical insults and work with the acid mantle and other substances secreted by the skin to ward off bacterial invasion. Finally, the skin acts as a biologic barrier by hosting elements of the immune system. Skin-resident dendritic cells act as a first line of defense, ingesting foreign substances, processing them, and presenting them to lymphocytes to activate the immune system. Dermal macrophages dispose of viruses and bacteria that penetrate the chemical and physical barriers of the skin and also act as antigen presenters (Marieb 2019).
Stress is formally defined as a state of threatened homeostasis provoked by a psychological, environmental, or physiologic stressor. While stress has considerable value in adapting to disturbances, in modern life it can be maladaptive, as shown by the fact that stress has been directly linked to risk for a broad range of diseases, including cardiovascular disease, metabolic disease, various psychiatric and neurodegenerative disorders, and cancer (Cohen 2007). Stress can be considered both an intrinsic and extrinsic factor that influences skin aging. Increasing evidence suggests there is a 2-way relationship between stress and skin quality, with direct impacts on keratinocyte and fibroblast number and function as well as on the composition of the extracellular matrix. It is also clear that psychological stressors lead to aberrant barrier function, with decreased epidermal lipid and structural protein synthesis, decreased hydration of the stratum corneum, and increased transepidermal water loss (Maarouf 2019), and can also result in immune system dysfunction leading to systemic and local inflammation (Liu 2017). Together, these factors interact to generate the visible and invisible signs of aging, make the skin more susceptible to environmental insult, and lower the threshold at which skin diseases manifest.
This white paper will not consider patients with skin diseases associated with a primary psychiatric disorder. These disorders, such as delusional infestation, somatic symptom disorders, and body dysmorphic disorder, are best managed in the setting of a mental health practice. Instead, here we will focus on the interaction between stressors and skin health. Lessons will be derived from what is known about the interaction between stress and psychological health in both healthy individuals and people with common dermatologic diseases, such as acne, psoriasis, rosacea, and atopic dermatitis. Finally, this white paper will examine the interaction between the dermatologist and the patient and the benefits for both in managing not only the skin but also the mind-skin connection.
The Link Between Skin Quality and Stress
It is well-established that stress exacerbates disease severity in patients with dermatologic diseases. For example, among patients with acne—the single most common skin condition seen by dermatologists—it is clear there is a strong relationship between stress and severity (Chiu 2003). After adjustment for change in sleep hours, perceived sleep quality, and perceived diet quality, increases in stress strongly correlated with a progressive increase in acne severity (r=0.61; P<0.01). Of note, stress experienced during an academic exam correlated with perceived acne severity, with patients who experienced intra-exam stress reporting a Leeds Acne Score that was approximately 30% higher than that reported during nonexamination periods (P<0.01). The relationship between stress and acne was further confirmed in a cross-sectional study conducted among 144 female medical students identified a strong relationship between stress severity and acne grade (P<0.01 for trend) (Figure 1) (Zari 2017). Similarly, stress ratings were directly associated with symptom severity in patients with rosacea or atopic dermatitis (Drummond 2017).
Figure 1. Relationship between stress severity and acne grade among female medical students (N=144). Acne grades ranged from 0 (no acne) to 3 (severe acne). Error bars: ±1.00 standard error (Zari 2017)

The connection between stressors and skin is perhaps best established in people with psoriasis, in whom stress has been associated with disease onset, flare-ups, and psychological distress. There is a 2-way interaction between stress and disease severity in many patients, as the perceived disfigurement and stigmatization of the disease results to anxiety, leading to a vicious cycle of increased stress and exacerbations in disease manifestations (Tampa 2018). In fact, more than two-thirds of patients with psoriasis report that stressful events have a direct association with disease flare (Xhaja 2014).
This link between stress and skin quality also exists in the general population who have not been diagnosed with underlying dermatologic conditions. For example, a study conducted in 529 medical students—who are at significant risk for severe stress—divided the study population into 3 groups: least stressed, moderately stressed, and highly stressed, based on the results of the validated Perceived Stress Questionnaire (PSQ) (Bin Saif 2018). Skin issues were measured using a self-reported skin complaints questionnaire. In this study, as compared with the least-stressed students, highly stressed students suffered from significantly more oily, waxy patches or flakes on the scalp, dry/sore rashes, warts, pimples, itchy skin, itchy hands, hair loss, scaly skin, troublesome sweating, and other rashes on the face (all P ≤ 0.05). The odds ratios for these conditions ranged from 1.94 for nail biting to nearly 5 for face rashes.
Mediators of The Mind-Skin Connection
Stress alters a number of pathways that can contribute to skin quality. Embryologically, the nervous system and the skin are derived from the same origin tissue: the ectoderm. As such, the brain and skin have some overlapping capabilities—for example, the skin has its own equivalent of the hypothalamic-pituitary-adrenal (HPA) axis, which may coordinate with the central HPA axis in the stress response.
Table 1 summarizes the major stress mediators in the skin, their sources, effector cells, and functions in skin. Each will be discussed in greater detail in this section.
Table 1. Major stress mediators in the skin. Adapted from Chen 2014 and references in the body of this white paper.
|
Stress Mediator |
Sources |
Skin Functions |
|
Corticotropin releasing hormone (CRH) |
·Hypothalamus ·Skin keratinocytes ·Sebacytes ·Mast cells |
·Stimulation of ACTH and cortisol production ·Proliferation, differentiation, apoptosis, inflammation, and angiogenesis |
|
Adrenocorticotropin (ACTH) |
·Pituitary gland ·Skin melanocytes, epidermal and hair ·Follicle keratinocytes and dermal fibroblasts ·Langerhans cells and macrophages |
·Stimulation of cortisol and corticosterone production ·Melanogenesis ·Cytokine production ·Cell proliferation ·Hair growth ·Immune regulation |
|
Cortisol |
· Adrenal cortex ·Skin hair follicles, melanocytes, and fibroblasts |
·Slows proliferation of epidermal keratinocytes ·Stimulates proliferation of fibroblasts and melanocytes ·Induces degranulation of mast cells ·Increases vascular permeability ·Induces expression of pro-inflammatory cytokines ·Compromises epithelial barrier function |
|
Neurotrophins |
·Central nervous system ·Skin sympathetic neurons ·Mast cells ·T and B cells ·Keratinocytes, fibroblasts, and melanocytes |
·Promote survival and differentiation of mast cells ·Modify inflammatory cytokines expression ·Promote proliferation of keratinocytes ·Important for melanocytes migration, viability, and differentiation ·Protect from oxidative stress and apoptosis ·Promote fibroblast differentiation and migration |
|
Substance P |
·Sensory nerve fibers |
·Induces inflammation by promoting cytokine release ·Activates mast cells ·Induces lymphocyte proliferation ·Induces vascular permeability ·May modulate the skin microbiota |
|
Prolactin |
·Pituitary gland ·Skin hair follicle and epidermal keratinocytes, fibroblasts ·Adipocytes ·Sweat glands ·Sebaceous glands |
·Stimulates keratinocyte proliferation and keratin production ·Stimulates sebum production in sebaceous glands ·May have an immunomodulatory role |
|
Catecholamines (epinephrine and norepinephrine) |
·Adrenal medulla ·Skin nerve fibers ·Keratinocytes |
·Regulate keratinocytes proliferation, differentiation, and migration ·Promote melanogenesis in melanocytes ·Decrease fibroblasts migration and collagen secretion ·Decrease wound healing |
The Central and Skin HPA Axis
The effect of stress on the skin is primarily mediated through the HPA axis. The sensation of stress is associated with the release of corticotropin-releasing hormone (CRH), which binds to its receptor on the pituitary gland and stimulates secretion of adrenocorticotropin (ACTH). ACTH, in turn, binds to receptors in the adrenal cortex, stimulating the production of glucocorticoids (GC) such as cortisol and corticosterone (Figure 2) (Marieb 2019).
Cortisol is the primary stress hormone in the human body. After binding to its receptor, a complex cascade of events results in changes in gene expression. Cortisol levels, under non-stressed conditions, naturally oscillate daily, with peak levels in the early morning and a nadir at around midnight. Stress can disrupt both the pattern and amount of cortisol produced, which can result in a relatively immunosuppressed state (Chen 2014).
Figure 2. A simplified representation of the HPA
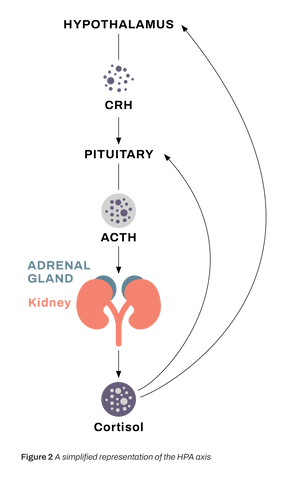
The skin itself has its own “HPA system” in that CRH and ACTH are produced by skin cells, which also express the cognate receptors for these molecules. In the skin, CRH is produced by keratinocytes, melanocytes, and mast cells in the presence of stressors such as immune cytokines, UV light, and cutaneous disease states. CRH has diverse effects in the skin, including slowing the proliferation of epidermal keratinocytes and stimulating proliferation of fibroblasts and melanocytes. CRH also induces degranulation of skin-resident mast cells, increases vascular permeability, and induces the production of the proinflammatory cytokine interleukin (IL)-6. ACTH stimulates production of IL-18, another pro-inflammatory cytokine (Chen 2014).
Skin barrier dysfunction is tightly associated with eczema, rosacea, psoriasis, acne, and other cutaneous diseases. However, variability in its health also contributes to skin quality in otherwise healthy patients. Much of the mind-skin connection is attributable to the overproduction of cortisol and its effect on the skin barrier. Increased cortisol production has been shown to slow the production of beneficial oils, resulting in dry, rough, irritated skin and accelerated transepidermal water loss. At the same time, cortisol stimulates the overproduction of sebum, increasing the likelihood and severity of acne. These changes also alter the pH of the skin, compromising the acid mantle and inducing changes in the composition of the skin’s commensal microbiota (Altemus 2001).
In a study that recruited 27 medical students without a dermatologic disease, levels of 11β-hydroxysteroid dehydrogenase 1, the enzyme that converts cortisone (the inactive form of cortisol) to cortisol, were measured and correlated with skin barrier function. In this study, elevated expression of 11β-hydroxysteroid dehydrogenase 1 in oral mucosa (which correlated closely with expression in epidermal keratinocytes) was associated with increased cortisol in the stratum corneum and deteriorated skin barrier function, as measured by decreased expression of keratinocyte differentiation markers.
The Skin and the SAM Axis
Stress is also associated with the release of catecholamines, such as epinephrine and norepinephrine, by modulating the activity of the sympathetic-adrenal medullary (SAM) axis (Chen 2014). These catecholamines are critical elements of the stress response and are involved in the fight-or-flight response to perceived danger. They have a broad range of well-understood effects, including heart rate and respiratory acceleration, vasoconstriction outside of muscles, and increased perspiration. In the epidermis, these signaling molecules downregulate keratinocyte proliferation, differentiation, and migration and increase melanogenesis in melanocytes. In the dermis, chronic epinephrine signaling appears to reduce fibroblast migration and collagen deposition—delaying wound healing—and also induces the expression of IL-6 (Romana-Souza 2011; Romana-Souza 2010). Signaling from these molecules has a less well-appreciated role in regulating the immune response; for example, data suggest that norepinephrine modulates human dendritic cell activation by altering cytokine release (Goyarts 2008).
Other Physiologically Active Stress Mediators
While the peripheral nervous system and the immune system are often considered wholly separate, abundant data suggest an intimate and bidirectional relationship (Chiu 2013). Both intrinsic factors—such as other stress mediators and inflammatory molecules—and extrinsic factors, such as noxious stimuli and microbial invasion, are detected by the peripheral nervous system, which responds via a number of secreted factors such as neuropeptides and neurotrophins. These factors amplify the local stress response and activate neurogenic inflammation (Chiu 2013).
Nerve growth factor, a member of the neurotrophin family, binds to cell-surface tyrosine kinase receptors expressed on mast cells, immune cells, keratinocytes, fibroblasts, and melanocytes (Chen 2014). Nerve growth factor is also secreted by immune cells and can act directly on peripheral sensory neurons to cause sensitization (Chiu 2013). Sensitization of nociceptors, in turn, results in increased release of neuropeptides that enhance immune cell activation, resulting in a positive feedback loop that drives the inflammatory response (Chen 2014).
Substance P, a member of the tachykinin neuropeptide family, is a stress-related neurotransmitter and neuromodulator that binds to receptors present on a broad range of cell types, including blood vessel endothelial cells, immune cells, fibroblasts, and neurons. Stress is associated with a significant increase in substance P-positive nerve fibers (Chen 2014). It has long been recognized as a modulator of pain perception, but it also plays roles in gastrointestinal function, memory, angiogenesis, vasodilation, and cell growth and proliferation (Graefe 2021). In the skin, substance P has been shown to play a role in initiating and enhancing the inflammatory response by stimulating cytokine release, activate mast cells, and induce lymphocyte permeability (Chen 2014).
Is There a Mind-Skin Microbiota Connection?
While the importance of the microbiota in gastrointestinal function is widely appreciated, the skin is also the home for a rich community of commensal and potentially pathogenic microorganisms, including bacteria, fungi, and viruses (Byrd 2018). Many of the normal microbial residents of the skin actively defend the body’s envelope by producing molecules that inhibit colonization of other microorganisms or alter their behavior. In healthy adults, the skin microbiota remains stable over time; however, increasing evidence suggests that stress can have a dramatic effect on the composition and behavior of these organisms. For example, glucocorticoids, which—as discussed above—are critical mediators of the stress response, have been shown to have a direct impact on the release of antimicrobial peptides by the skin (Holmes 2015).
In addition to its direct roles in neuroinflammation, substance P also appears to provide a link between stress and the microbial flora of the skin (Mijouin 2013). It is released in sweat during stress and has both direct and indirect antimicrobial activity by acting as an antimicrobial peptide and stimulating the release of cathelicidins and defensins. However, certain bacteria, such as Bacillus cereus—a common transient resident of the skin—can detect and respond to substance P in a manner that increases virulence. In the case of B cereus, substance P induces the release of superoxide dismutase, an enzyme that protects the bacteria against oxidative stress, increases bacterial production of collagenase, and stimulates the formation of protective biofilms (Mijouin 2013). Similarly, both Staphylococcus aureus and Staphylococcus epidermidis respond to the presence of substance P with a marked increase in cytotoxicity (Mijouin 2013). Data also suggest that cortisol significantly increases the inflammatory response to Propionibacterium acnes, the bacterium associated with the pathogenesis of acne and several other opportunistic infections (Holmes 2015).
What is the Impact of Stress on Inflammation?
The balance between pro- and anti-inflammatory factors is altered by stress. The net effect of stress on inflammatory mediators changes with the chronicity of exposure to stress. Acute stress is associated with an adaptive response that enhances immune response, whereas chronic stress often results in immunosuppression. Intense stress may overactivate the immune system, resulting in the release of a variety of pro-inflammatory mediators including C-reactive protein, IL-6, TNFα, IL-1β, and activate the transcription factor nuclear factor kappa B (NF-κB), which is known to play a role in modulating the expression of pro-inflammatory products (Liu 2017).
The effects of stress are mediated, in part, by glucocorticoids released by the adrenal glands during the stress response (discussed above). Glucocorticoids have a range of effects on the immune system that depending on the duration of release, have anti- or pro-inflammatory effects. As noted above, stress is a clear trigger for many inflammatory diseases of the skin, such as psoriasis, thus providing a direct clinical link between stress, inflammation, and symptoms.
What is the Impact of Stress on Skin Aging?
The data summarized above suggest the presence of a direct mind-skin connection and that stress has potentially detrimental effects that are mediated by multiple intimately interconnected pathways. It is also well understood and readily apparent that chronic stress is a major contributor to the appearance of aged skin; this connection may be mediated both through the neuroendocrine and immune systems (Dunn 2013; Liu 2017). Telomere shortening, leading to premature replicative senescence of skin cells, may also provide a link between stress and skin aging, although the precise underlying mechanisms are not yet fully elucidated (Chen 2014).
Continuous, low-grade inflammation—such as that induced by chronic stress—may cause gradual tissue damage that, while present systemically, manifests visibly in skin aging (Zhuang 2014). This has been referred to by some authors as “inflammaging.”
The effects of inflammation on the skin can be modeled through experimental exposure of skin to ultraviolet light, which induces epidermal keratinocytes to release inflammatory cytokines, induces mast cells to generate prostaglandins and other inflammatory mediators, and results in the massive infiltration of neutrophils into the epidermis and dermis, where they play roles in clearing apoptotic cells. However, these cells also indiscriminately release a number of degradative enzymes, such as neutrophil elastase and various matrix metalloproteinases, that degrade the extracellular matrix. The complement system is also induced under conditions of stress and can recruit and activate dermal macrophages. Table 2 summarizes the role of inflammatory factors known to have a role in skin aging.
Table 2. Known key inflammatory factors and their role in skin aging (Zhuang 2014)
|
Proinflammatory Factors |
Pathway and Potential Skin Damage |
|
Reactive oxygen species |
Cause skin cell damage; generate oxidized lipids; induce MMP expression in dermal fibroblasts |
|
TNFα, IL-1 |
Initiate inflammatory responses in skin and induce synthesis and release of other pro-inflammatory cytokines |
|
IL-6, IL-8, etc |
Recruit neutrophils and macrophages; activate dermal fibroblasts to secrete MMPs |
|
Neutrophils |
Produce elastase and MMPs that cause ECM degradation |
|
MMPs |
Cause ECM degradation, damage dermal connective tissue and cause visible skin aging |
|
Complement system |
Activates macrophages; induced particularly by UV radiation |
|
Macrophages |
Infiltrate skin after UV exposure; generate ROS and MMPs that cause ECM degradation |
ECM = extracellular matrix; IL = interleukin; MMP = matrix metalloproteinase; ROS = reactive oxygen species; TNF = tumor necrosis factor; UV = ultraviolet.
Of note, poor sleep quality—which is intimately connected to stress—is likely to contribute to skin aging. The skin of individuals who suffer from poor quality sleep is marked by relatively reduced elasticity, fine lines, uneven pigmentation, and slow recovery from skin barrier disruption (Oyetakin-White 2013). Stressors such as smoking and air pollution are both considered contributors to premature skin aging. The effects of these environmental stressors may be mediated by an increase in reactive oxygen species (ROS) production, vitamin E depletion, lipid peroxidation, and the induction of collagen- and elastin-degrading matrix metalloproteinase expression (Chen 2014; van Doren 2015).
Can Stress Reduction Interventions Improve Skin Quality?
Direct Evidence for Stress Reduction and Improvements in Skin Quality
The majority of evidence for a link between stress reduction and skin quality comes from studies in patients with psoriasis and atopic dermatitis.
The impact of stress reduction through mindfulness meditation has been prospectively evaluated in patients with psoriasis (Kabat Zinn 1998). In this study, patients were randomly assigned to either a mindfulness meditation guided by audiotaped instructions during phototherapy or photochemotherapy, or a control condition of the light treatments alone without meditation instructions. Patients who received mindfulness meditation instruction were significantly more likely to reach 50% skin clearance (P = 0.013) and complete skin clearance (P = 0.033). Similarly, a small case-control study of relaxation therapy found profound differences in clearing among patients with psoriasis: among patients who were trained in relaxation therapy, 70% achieved Psoriasis Area Severity Index (PASI) 50 at the end of 2 months, as compared with 13.3% of the control group (Neerackal 2020).
The efficacy of stress-reduction modalities have also been explored in atopic dermatitis. In one trial, patients (N=102) with atopic dermatitis were randomly allocated to 12 weeks of cognitive-behavioral therapy (CBT), delivered by internet, or a control that gave subjects about standard care; the primary outcome measure was the difference in atopic dermatitis symptoms, as measured by the Patient-Oriented Eczema Measure (Lagerlof 2021). At 12 weeks, patients who received CBT had significant, moderate to large reductions in atopic dermatitis symptoms, combined with significant reductions in itch intensity, perceived stress, sleep problems, and depression. Of note, these gains were maintained through 12 months. A second study found that psychological interventions in patients with atopic dermatitis were associated with significantly larger improvements in skin conditions than standard medical care and were associated with significant reductions in the need for topical steroid treatment (Ehlers 1995).
Aerobic exercise has broad, systemic benefits in the human body, including reduction of stress, and data suggest that these benefits can also be extended to the skin. For example, in one recent study conducted in humans, short-term aerobic exercise was associated with reduced signs of skin aging, including a reduction in stratum corneum thickness (which generally increases in thickness with age), increased stratum spinosum thickness in elderly subjects, and increased reticular dermis content. These effects may be mediated by increases in circulating IL-15, an exercise-induced hormone that has previously been associated with reduced adiposity, increased muscle mass, improved mitochondrial function and exercise capacity (Crane 2015).
Finally, limited data suggest that aromatherapy may have some benefit (with the caution that the aerosolized oils may exacerbate skin conditions in sensitive individuals). In one study, aromatherapy was associated with a reduction in stress-induced serum and salivary cortisol and enhanced skin barrier recovery, as measured by transepidermal water loss, in both animal models and humans (Fukuda 2012).
Other Stress-Reduction Modalities
Mindfulness is a popular meditation technique that consists of 2 parts: attention and acceptance. Attention involves “tuning in” to experiences to focus on what is happening in the present moment, while acceptance involves observing those feelings and sensations without judgment (American Psychiatric Association 2021). One case-control study and one randomized clinical trial with reasonable methodologic quality showed that meditation was associated with increased telomere length; this effect was significant in female meditators (Desanayaka 2021). The effect of meditation on stress markers, such as cortisol, has been better studied. For example, in one meta-analysis of 10 studies using blood samples, meditation interventions had a moderate, albeit statistically significant, effect on cortisol levels. This effect was primarily seen in subjects with existing somatic illnesses, such as type 2 diabetes mellitus (Koncz 2021).
A number of additional stress-reduction modalities may be viable candidates to consider for improving overall stress. Yoga, as it is practiced widely worldwide, has been shown to impact both the neuroendocrine and immune systems. In 1 meta-analysis of 15 relatively high-quality studies, yoga was shown to reduce inflammatory markers, including IL-6, C-reactive protein, and tumor necrosis factor, with a distinct dose-response relationship (Djalilova 2019). Yoga has also been shown to be associated with reliable reductions in serum cortisol, the primary stress hormone in the human body (Katuri 2016; Cahn 2017). In 1 reasonably well-conducted study in 121 healthy individuals, guided relaxation was associated with improved skin barrier recovery following induction of mild skin damage by tape stripping (Robinson 2015). Similarly, some evidence suggests that rituals may have a potentially anxiolytic effect by reducing cognitive load (Karl 2018).
Conclusions
Dermatologists have long accepted the role of stress in skin health, as it is readily apparent in the clinic in otherwise healthy individuals. Abundant evidence exists that stress and skin function and quality are intimately linked, and some evidence suggests that interventions to reduce stress, such as mindfulness meditation, cognitive-behavioral therapy, yoga, exercise, and even aromatherapy may have a positive impact on skin quality. Rituals, such as taking time with the daily application of cosmetic products, also may have the side benefit of decreasing psychological stress responses.
There is an underappreciated opportunity in the dermatology clinic to improve outcomes and increase patient satisfaction with their overall treatment plan by providing guidance on stress management through non-medical interventions. From a practical perspective, the dermatologist’s time to address the sometimes complex factors underlying stress is limited; thus, there may be a role for partnerships with outside services that specialize in helping patients manage stress.
Additional Reading
For those with a particular interest in the mind-skin connection, we recommend the following reviews.
Chen Y, Lyga J. Brain-skin connection: Stress, inflammation, and skin aging. Inflamm Allergy—Drug Targets. 2014;13:177-190.
Chiu A, Chon SQY, Kimball AB. The response of skin disease to stress. Arch Dermatol. 2003;139:897-900.
Holmes CJ, Plichta JK, Gamelli R et al. Dynamic role of host stress responses in modulating the cutaneous microbiome: implications for wound healing and infection. Adv Wound Care. 2015;4:24-37.
Maarouf M, Maarouf CL, Yosipovitch G et al. The impact of stress on epidermal barrier function: an evidence-based review. Br J Dermatol. 2019;181:1129-1137.
References
Altemus M, Rao B, Dhabhar FS et al. Stress-induced changes in skin barrier function in healthy women. J Invest Dermatol. 2001;117:309-317.
Bin Saif GA, Alotaibi HM, Alzolibani AA et al. Association of psychological stress with skin symptoms among medical students. Saudi Med J. 2018;39:59-66.
Byrd AL, Belkaid Y, Segre JA. The human skin microbiome. Nat Rev Microbiol. 2018;16:143-155.
Cahn BR, Goodman MS, Peterson CT et al. Yoga, mediation, and mind-body health: Increased BDNF, cortisol awakening response, and altered inflammatory marker expression after a 3-month yoga and meditation retreat. Front Hum Neurosci. 2017.
Chen Y, Lyga J. Brain-skin connection: Stress, inflammation, and skin aging. Inflamm Allergy—Drug Targets. 2014;13:177-190.
Chiu A, Chon SQY, Kimball AB. The response of skin disease to stress. Arch Dermatol. 2003;139:897-900.
Chiu IM, von Hehn CA, Woolf CJ. Neurogenic inflammation – the peripheral nervous system’s role in host defense and immunopathology. Nat Neurosci. 2013;15:1063-1067.
Choe SJ, Kim D, Kim EJ et al. Psychological stress deteriorates skin barrier function by activating 11β-hydroxysteroid dehydrogenase 1 and the HPA axis. Sci Rep. 2018;8:6334.
Cohen S, Janicki-Deverts D, Miller GE. Psychological stress and disease. JAMA. 2007;298:1685-1687.
Crane JD, MacNeil LG, Lally JS et al. Exercise-stimulated interleukin-15 is controlled by AMPK and regulates skin metabolism and aging. Aging Cell. 2015;14:625-634.
Desanayaka NN, Sirisena ND, Smaranayake N. The effects of meditation on length of telomeres in healthy individuals: A systematic review. Systematic Rev. 2021;10:1-6.
Djalilova DM, Schulz PS, Berger AM et al. Impact of yoga on inflammatory biomarkers: A systematic review. Biol Res Nurs. 2018;21:198-209.
Drummond PD, Su D. Increases in psychological stress precede flares of rosacea: A prospective study. J Clin Exp Dermatol Res. 2017;8:1-4.
Dunn JH, Koo J. Psychological stress and skin aging: A review of possible mechanisms and potential therapies. Dermatol Online J. 2013;19:18561.
Ehlers A, Stangier U, Gieler U. Treatment of atopic dermatitis: A comparison of psychological and dermatological approaches to relapse prevention. J Consult Clin Psychol. 1996;63:624-625.
Fukada M, Kano E, Miyoshi M et al. Effect of ‘rose essential oil’ inhalation on stress-induced skin-barrier disruption in rats and humans. Chem Senses. 2012; 37:347-356.
Goyarts E, Matsui M, Mammone T et al. Norepinephrine modulates human dendritic cell activation by altering cytokine release. Exp Dermatol. 2008;17:188-196.
Graefe S, Mohiuddin SS. Biochemistry, substance P. StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing 2021.
Hedman-Lagerlof E, Fust J, Axelsson E et al. Internet-delivered cognitive behavior therapy for atopic dermatitis: A randomized clinical trial. JAMA Dermatol. 2021;157:796-804.
Holmes CJ, Plichta JK, Gamelli R et al. Dynamic role of host stress responses in modulating the cutaneous microbiome: implications for wound healing and infection. Adv Wound Care. 2015;4:24-37.
Neerackal RJ, Latheef NA, Sukumarakurup S, Jaffernay M. Relaxation therapy in the management of psoriasis. Derm Ther. 2020;33:e14030.
Kabat-Zinn J, Wheeler E, Light T et al. Influence of mindfulness meditation-based stress reduction intervention on rates of skin clearing in patients with moderate to severe psoriasis undergoing phototherapy (UVB) or photochemotherapy (PUVA). Psychosomatic Med. 1998;60:625-632.
Karl JA, Fischer R. Rituals, repetitiveness, and cognitive load: A competitive test of ritual benefits for stress. Hum Nat. 2018;29:418-441.
Katuri KK, Dasari AB, Kurapati S et al. Association of yoga practice and serum cortisol levels in chronic periodontitis patients with stress-related anxiety and depression. J Int Soc Prev Community Dent. 2016;6:7-14.
Kinsinger SW. Cognitive-behavioral therapy for patients with irritable bowel syndrome: current insights. Psychol Res Behav Manag. 2017;10:231-237.
Koncz A, Demetrovics Z, Takacs ZK. Meditation interventions efficiently reduce cortisol levels of at-risk samples: A meta-analysis. Health Psych Rev. 2021;15:56-84.
Liu Y-Z, Wang Y-X, Jiang C-L. Inflammation: The common pathway of stress-related diseases. Front Hum Neurosci. 2017;11:316-326.
Maarouf M, Maarouf CL, Yosipovitch G et al. The impact of stress on epidermal barrier function: an evidence-based review. Br J Dermatol. 2019;181:1129-1137.
Marieb EN, Hoehn K. Human Anatomy & Physiology. 11th ed. Pearson Educational Ltd. 2019.
Mijouin L, Hillion M, Ramdani Y et al. Effects of a skin neuropeptide (substance P) on cutaneous microflora. PLOS One. 2013;8:e78773.
Oyetakin-White P, Koo B, Matsui M et al. Effects of sleep quality on skin aging and function. J Invest Dermatol. 2013;S126.
Robinson H, Jarrett P, Broadbent E. The effects of relaxation before or after skin damage on skin barrier recovery: A preliminary study. Psychosom Med. 2015;77:844-852.
Romana-Souza B, Otranto M, Almeida TF et al. Stress-induced epinephrine levels compromise murine dermal fibroblast activity through beta-adrenoreceptors. Exp Dermatol. 2011;20:413-419.
Romana-Souza B, Otranto M, Vieira AM et al. Rotational stress-induced increase in epinephrine levels delays cutaneous would healing in mice. Rain Behav Immunol. 2010;24:427-437.
Sandoval MHL, Ayres EL. Skin aging and stress. Stress and Skin Disorders. Springer International Publishing, Switzerland. 2017.
Tampa M, Sarbu M-I, Mitran M-I. The pathophysiological mechanisms and the question for biomarkers in psoriasis, a stress-related skin disease. Disease Markers. 2018;5823684.
van Doren SR. Matrix metalloproteinase interactions with collagen and elastin. Matrix Biol. 2015;224-231.
Zari S, Alrahmani D. The association between stress and acne among female medical students in Jeddah, Saudi Arabia. Clin Cosm Invest Dermatol. 2017;10:503-506.
Zhuang Y, Lyga J. Inflammaging in skin and other tissues – the role of complement system and macrophage. Inflamm Allergy Drug Targets. 2014;13:153-161.




Leave a comment
This site is protected by hCaptcha and the hCaptcha Privacy Policy and Terms of Service apply.